THE ELECTRON
650 likes | 842 Views
THE ELECTRON. Part 1. Electrons and Quantum Theory. Ever wondered where the colors come from in fireworks or neon lights? The explanation for these colors is tied up in e - s and atoms and energies

THE ELECTRON
E N D
Presentation Transcript
THE ELECTRON Part 1
Electrons and Quantum Theory • Ever wondered where the colors come from in fireworks or neon lights? • The explanation for these colors is tied up in e-s and atoms and energies • Niels Bohr proposed his model in part to explain that most elements particularly metals and gases tend to glow with a colored light when heated or electrified.
Electrons and Quantum Theory • As we discussed earlier, e-s are on energy levels of quantized energy • If the e-s are excited they are forced to occupy levels of higher energies • As the e-s lose the energy they absorbed they fall back to their original level (ground state), which means they lose energy, sometimes this energy is visible (colored light).
Electrons and Quantum Theory • It turns out that the colored light is a mixture of quantized light energies. • A quantum of light is called a photon • There are several versions of light energy, and they all have 1 common characteristic…speed. • Light is a special type of non-ionizing radiation called electromagnetic radiation
The Behavior of Light • The EM spectrum is a broad range of wavelengths of energy which are all classified together because of their common speed. • Each of the different types of EMR all travel through space (vacuum) at a speed just under 300,000,000 m/s. • Radio, x-rays, ultraviolet, infrared, microwaves, etc. are versions of EMR
Light as a Wave • All waves, can be described in terms of 4 characteristics • Frequency • Wavelength • Amplitude • Speed Distinguishes one type of wave from another (sound, water, EMR, etc.) Define one EMR from another
Light as a Wave • Wavelength (l): • the distance between successive crests of the wave. • the distance that the wave travelsasit completes one full cycleof up and down motion
Light as a Wave • Frequency (): • How fast the wave oscillates. • Measured by the # of times a light wave completes a cycle of up and down motionper sec. • When a radio station identifies itself it’s the frequency used • Unit is a Hertz (sec-1)
Light as a Wave • Amplitude: • Is theheight of the wavemeasured from the origin to its crest, or peak • Thebrightness, orintensityof light depends on the amplitude of the light wave. amplitude
Light as a Wave • Speed (c): • Regardless of its wavelength, each type of EMR moves through space at a constant speed • 3.00x108 m/s • Nothing can go faster than light, it’s the fastest thing ever (in a vacuum) • Light can be slowed down as it passes through air, water, glass, etc.
speed (frequency) (wavelength) = Light as a Wave • Since light moves at a constant speed there is a mathematical relationship between frequency () & wavelength () • Theshorterthe wavelength thehigherthe frequency • Thelongerthe wavelength the lower the frequency c =
Example: Orchestras in the United States tune their instruments to an "A" that has a frequency of 440 cycles per second, or 440 Hz. If the speed of sound is 1116 feet per second, what is the wavelength of this note? c = 1116 ft/sec = (440 sec-1) = 2.5 ft
Example 2: Calculate the frequency of red light that has a wavelength of 700.0 nm c = Speed of light = 3.00 x 108 m/sec 3.00 x 108 m/sec = (7.000 x 10-7 m)() = 4.286 x 1014 sec-1
Your Turn: • A very bright yellow line in the emission spectrum of sodium has a frequency of 5.10 x 1014 Hz (5.10 x 1014 s-1). Calculate the wavelength of this yellow light. • What frequency is radiation with a wavelength of 5.00 x 10-6 cm? In what region of the electromagnetic spectrum is this radiation?
Light as a Wave: White Light • As scientists strived to learn more about light, they discovered that white light (sunlight) is a mixture of 7 colors • Remember, white light encompasses only the visible portion of the spectrum • It is a mixture which can be separated by a prisminto a continuous spectrum
Light as a Wave: White Light • The colors that combine to form white light are red, orange, yellow, green, blue, indigo, and violet (ROYGBIV) • The different colors have different wavelengths and frequencies • Shortest & highest =violet • Longest & lowest =red
Elements as Light • Scientists soon discovered that elements can also produce light. • If you energize gaseous elements they glow with a characteristic colored light • Neon glows orange, strontium glows red, copper glows green, etc.
Light as a Wave • If you take elemental light and pass it through a prism the light does not produce a continuous spectrum • Instead the spectrum splits into a characteristic pattern of lines of color. • It’s not a mixture of all wavelengths, but a mixture of specific,individual wavelengths • For instance with Hydrogen, you see 4 distinct lines of color
Quantum Theory • Before Max Planck came up with the model of quantized energy, scientists had no idea why excited elements glowed with light that was a mixture of specific wavelengths and not broad spectrums of wavelengths • If energy is lost or gained in discrete bundles with specific energy this would explain why we see individual lines of specific colors no matter how complex the spectra
Quantum Theory • Planck suggested that energy, instead of being given off in continuous waves, is instead given off in little packets of energy, or quanta. • The word quantum means afixed amount, think of it as flashes of energy • Also calleda photon when describing a quantum of light
Quantum Theory • Planck’s idea was that one quantum of energy (light) was related to its frequency by the equation: E = h • The constant h (planck’s constant) has a value of 6.6262 x 10-34 J-s, E is the energy, and is the frequency of the radiation. • The energy in wave form that is abs-orbed or emitted by atoms, is restrict-ed to specific quantities (quantized)
Example: How much energy does a photon have that has a 700.0 nm wavelength? E = h We determined the frequency in a previous example: = 4.286 x 1014 sec-1 E = (6.626 x 10-34J•sec)(4.286 x 1014 sec-1) E = 2.840 x 10-19 J
Your Turn: • When an electron falls from the fourth to the second energy level, it emits a photon of green light with a frequency of 5.80 x 1014 s-1. Calculate the energy of this photon. • A photon of red light has a wavelength of 645 nm. Calculate the energy of this photon.
Quantum Theory • Planck’s understanding works because of the size of planck’s constant (h). • Each quantum (leap) is10-34, so it feels like a continuous change of energy at the macroscopic level • Just like a drawn line with a computer looks smooth unless you zoom in to see it is actually blocks
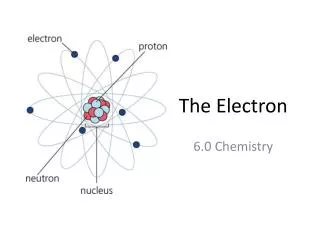
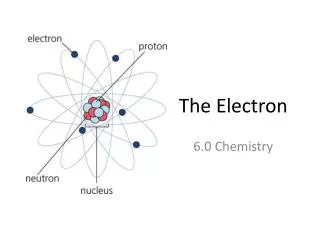
Electrons and Quantum Theory • So how does this all relate to the atom and the electrons in the atom? • Remember Bohr reasoned that e-s existed on orbits of quantized energy around the nucleus. • Every energy level (n) could contain a maximum number of e-s having that amount of energy • If every e- in the atom has its minimum amnt of energy it’s in its ground state
Electrons and Quantum Theory • If an e- was to gain energy it would then have too much energy to remain on its particular level with its particular energy • The atom is in an excited state • The excited e- leaps to a higher energy level that allows its kind of energy. • An e- doesn’t stay excited, it eventually loses the energy it gained • The excited e- will return to ground state by releasing its absorbed energy all at once, or in combination
Electrons and Quantum Theory • The energy released will be a photon or multiple photons, of specific frequencies • If that fall is back to n=2 the photon will be a photon in the visible range. • The more energy absorbed by the e- the higher the leap in energy (the further away from the nucleus) • The higher the leap - the farther the electron has to fall back down • Each level it falls a specific wavelength with a specific frequency is emitted; therefore specific lines of color
Electrons and Quantum Theory • Bohr used his theory to calculate the frequencies & wavelengths emitted by excited H atoms accurately • which was powerful evidence in support of his model. • However, it only worked successfully for Hydrogen
Quantum Theory • Even Einstein dabbled in quantum physics. • Albert Einstein saw the potential of quantized energy and proposed it to be a new way of understanding light. • He needed Planck’s work to explain his Nobel Prize winning research on the photoelectric effect.
Photoelectric Effect • Scientists had noticed that when you shine light onto some types of metal, a measurable voltage is produced • The light seems to transfer energy to the metal which causes an electric current • But, not every kind of light produces the current • And it doesn’t help to initiate the current by making the light brighter
Photoelectric Effect • For each metal, a minimum frequency of light is needed to release e- • Red light cannot produce a current • butvioletcan produce a current
Photoelectric Effect • Einstein hypothesized that since light exists as quantized energy, the bundle of energy can behave much like a billiard ball • Each packet/photon acts as a particle as it collides with an e- in the metal • If it has sufficient energy it can kick the e- completely out of the atom, which produces an electric current
Photoelectric Effect • Einstein reasoned that the frequency of the photon determines whether or not it has sufficient energy to eject an e- • There is a minimum frequency of light required to establish a current • This is why higher energy forms of light can do damage to organisms • they can knock electrons out of the atoms in our cells causing chemical bonds to be broken, possibly causing irreparable damage
Wave…I mean…Particle…I mean… • Light exhibits the properties of both particles and waves. • Light can be thought of as a tiny ball which can collide with an electron • This is known as the dual nature of light • It stands to reason since the e- is a particle that has quantized energy it would also have a frequency and wavelength just like light does. • This is known as the dual nature of the e-
Matter = Wave & Particle • If light acts like energy and like matter, maybe matter can interact like both too • This connection was first made by Louis de Broglie • Louis de Broglie reasoned that matter, or specifically, the electron can behave like a standing wave and at times exhibit the characteristics of a wave, much like light.
Matter = Wave & Particle • He developed a relationship between the mass & velocity of a particle and the wavelength it would exhibit • = h/mv. • Which predicts that all objects in motion has wavelike characteristics • it is only noticeable in objects with a tiny mass. • The electron should be extremely predictable then, according to classical physics, but…
The Quantum Model of the Atom • In 1927, Werner Heisenberg proposed the uncertainty principle. • It states, that you can’t know both the velocity & momentum of a particle simultaneously • So we can’t know the exact location of an e- or it’s path in the atom, & any attempt to measure the velocity will influence the momentum & vice versa