Fixed Bed Reactor
720 likes | 3.89k Views
Fixed Bed Reactor. Quak Foo Lee Chemical and Biological Engineering The University of British Columbia. C A,out. Regeneration. 1. 2. C A,in. Fixed Bed Reactor. Solids take part in reaction unsteady state or semi-batch mode Over some time, solids either replaced or regenerated.

Fixed Bed Reactor
E N D
Presentation Transcript
Fixed Bed Reactor Quak Foo Lee Chemical and Biological Engineering The University of British Columbia
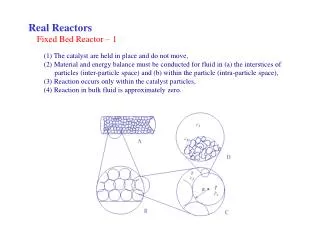
CA,out Regeneration 1 2 CA,in Fixed Bed Reactor • Solids take part in reaction unsteady state or semi-batch mode • Over some time, solids either replaced or regenerated Breakthrough curve CA,out/CA,in t
Isothermal Reaction:Plug Flow Reactor • Plug flow of fluid – no radial gradients, and no axial dispersion • Constant density with position • Superficial velocity remains constant
Plug Flow Model z + dz CA,f + dCA,f z CA,f U0 (m/s) superficial velocity
Mass Balance Input – Output – Reaction = Accumulation Divide by ∂z and take the limits as ∂z 0 ε is void fraction in bed
Void fraction For first order reaction, fluid only: For steady state: Volume of reactor Therefore,
Conversion as a function of Height Integrating with CA,f = CA,f,in at z = 0 Note 1: Same equation as for catalytic reactor with 1st order reaction Note 2: Can be used in pseudo-homogeneous reaction
Balance on Solid • aA (fluid) + S (solid) Products • Input – Output – Reaction = Accumulation • Over increment of dz: input = 0, output =0 Volume fraction of solid = m3 of solid m3 of reactor volume mol m3 of solid · s
Solve These Equations = 0 (In quasi steady state, we ignore the accumulation of A in gas) Substitute rAv
Non-Isothermal Packed Bed Reactor • For mass continuity did balance on fluid and on solid • For energy balance, we do balance on each phase
Non-Isothermal Packed Bed Reactor • Assumptions: • Adiabatic reaction – no heat lost through shell to surroundings (no radial temperature gradients) q = 0 • Biλ is small – uniform T within particle (an exothermic reaction Tp > Tg) • Plug flow of gas and use Tref =0 for enthalpy calculations • Assume an average density can be used (ρg = constant)
Modeling q =0 Tf + dTf z + dz Tf z Tf,0 U0
Governing Equations Solve all these equations together. • Fluid • Solid Quasi Steady State
For Heat Transfer • Z=0 all t Tf = Tf,0; Ts = Ts,0 • T = 0 all z Ts = Ts,0; Tf = Tf,0 • Generally, require a numerical solution for the solution of the set of equations.