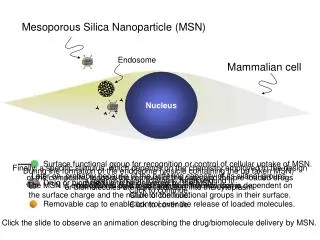
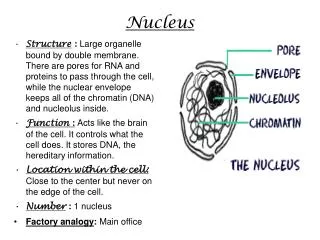
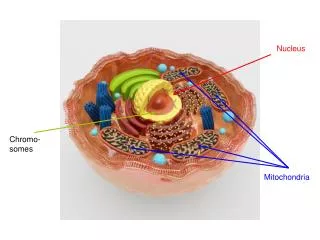
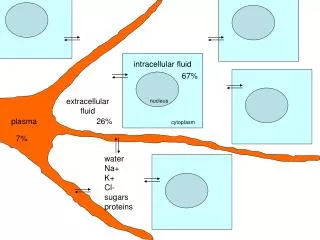
Nucleus
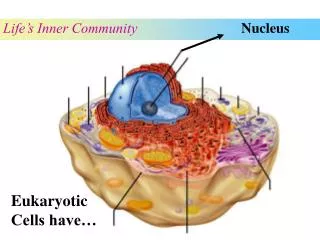
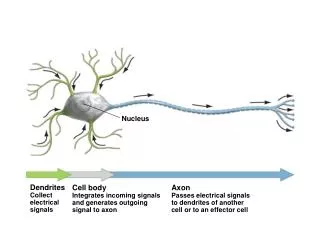
Nucleus. The nucleus consists of protons and neutrons. Protons: Z ( atomic number ) Neutrons: N Nucleons: A = Z + N ( atomic mass ) Full notation shows A, Z Chemical symbol duplicates atomic number. Short notation – A only. Nuclear Notation.

Nucleus
E N D
Presentation Transcript
The nucleus consists of protons and neutrons. Protons: Z (atomic number) Neutrons: N Nucleons: A = Z + N (atomic mass) Full notation shows A, Z Chemical symbol duplicates atomic number. Short notation – A only Nuclear Notation
All nuclei have about the same density r. Independent of A The nucleus is roughly a sphere. Radius r, r0= 1.2 fm Volume V Nuclear Size
Nuclei that have the same proton count are isotopes. Isotones for identical neutron count Isotopes
Energy measurements for nuclear physics are built on the electron volt (eV). 1 eV = 1.6 10-19 J Mass is expressed in terms of the rest energy. Mass is also expressed in terms of nucleons. Atomic mass unit (u) Based on 12C 1 u = 931.5 MeV/c2 Proton, p 938.3 MeV/c2 1.007 u Neutron, n 939.6 MeV/c2 1.009 u Electron, e 0.511 MeV/c2 5.546 10-4 u Atomic Mass
The mass (M) in u is nearly equal to the atomic number (A). Tables of isotope data frequently list D = M – A. Binding energy 1H; D = 7.29 MeV 4He; D = 2.42 MeV 56Fe; D = – 60.60 MeV 214Pb; D = – 0.15 MeV 218Po; D = 8.38 MeV 222Rn; D = 16.39 MeV 226Ra; D = 23.69 MeV Mass Difference
Binding Energy next