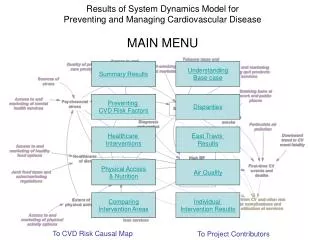
Results:
In Vitro Biocompatibility of Iron Filled Carbon Nanotubes Taylor, A. 1 , Krämer, K. 1 , Hampel, S. 2 , Füssel, S. 1 , Klingeler, R. 2 , Ritschel, M. 2 , Büchner, B. 2 , Wirth, M. P. 1

Results:
E N D
Presentation Transcript
In Vitro Biocompatibility of Iron Filled Carbon Nanotubes Taylor, A.1, Krämer, K.1, Hampel, S.2, Füssel, S.1, Klingeler, R.2, Ritschel, M.2, Büchner, B.2, Wirth, M. P.1 1. Department of Urology, Faculty of Medicine, Dresden University of Technology, Germany 2. Leibniz Institute for Solid State and Materials Research (IFW) Dresden, Germany. http://urologie.uniklinikum-dresden.de / arthur.taylor@uniklinikum-dresden.de Research was supported in part by the European Network CARBIO, Contract MRTN-CT-2006-035616. • Introduction & Objectives: • Magnetic Fluid Hyperthermia is a potential therapy for treating cancer. • Most of current research is focused on nanoparticles made of iron oxides since other magnetic materials contain potentially toxic elements. • Metallic iron could enhance the heating effect. Its application is however hindered because of oxidation in ambient or biological conditions. • Carbon nanotubes (CNTs) can overcome this problem, as a protective coating for iron. • Furthermore, they could be explored as multi-functional containers carrying not only iron but other materials and/or molecules of interest (Figure 1). • Aim of this study: explore the biocompatibility of iron-filled carbon nanotubes in comparison to empty ones. Results: Results: 1,8 µm (a) (c) 1,8 µm (e) 400 × CNT Wall Figure 5. Cell cycle distribution of PC-3 cells after exposure to iron filled CNTs as evaluated by PI staining and FACS analysis. Iron Filling 1,8 µm (d) (b) (f) 0,08 µm 400 × Figure 2. Association of iron filled nanotubes with cells as observed by optical and transmission microscopy. Control PC-3 cells (a) and cells exposed to 100 µg/ml of iron filled CNTs for 4h (b). Control EJ28 cells (c) and exposed to 100 µg/ml of empty (d) or iron filled carbon nanotubes (e) for 24h. Higher magnification of iron filled CNT inside of EJ28 cell (f). CNT filling (magnet, temperature sensor, drugs, ...) Targeting/Solubilizing moieties • CNTs were found internalized by cells and also well attached to the cellular membrane. Results: Figure 1. Schematic representation of a multi-functional nanocontainer. • Materials & Methods: • Iron filled carbon nanotubes were synthesized by catalytic decomposition of ferrocene in a CVD system and characterized by TEM, X-Ray diffraction and SQUID magnometry. • In order to suspend the nanotubes in aqueous solutions, an ultrasonication procedure in a PBS solution containing albumin in a 1:1 weight ratio in respect to the nanotubes was applied. • Cellular uptake was evaluated by transmission electron microscopy in bladder (EJ28) and prostate cancer cells lines (PC-3) and non-malignant fibroblasts. • Toxicity was studied in a time (24h, 48h and 96h) and concentration dependance (6.2, 25 and 100 µg/ml) using a prostate cancer cell line (PC-3) and a non-malignant cell lineage (fibroblasts). • Effect of nanotubes was evaluated by measurement of: • metabolic activity (WST-1 assay), • cell proliferation (cell counting), • apoptosis rate (Annexin-V/PI labelling and FACS analysis) and • cell cycle distribution (PI staining and FACS analysis). Figure 6. Apoptosis rate of PC3 cells after exposure to iron filled CNTs as evaluated by Annexin-V/PI labelling and FACS analysis. • A small concentration dependant decay in viability was observed 24h after incubation with iron filled carbon nanotubes. After 48h, viability was recovered and remained stable up to 72h, when cells reached confluency. • Cell proliferation was not impaired by incubation with empty nor filled nanotubes. • Cell cycle distribution was identical to control conditions, indicating no adverse effects from the presence of nanotubes. A decrease in the number of cells in the S and G2 phase is observed because confluence is reached. • The number of apoptotic cells was comparable to control conditions in all cases, indicating no toxic effects induced by nanotubes. • Similar results obtained with fibroblasts, indicating biocompatibility with malignant and non-malignant cell lines. Figure 3. Relative viability of PC-3 cells after exposure to empty and iron filled CNTs as evaluated by WST-1 assay. • Conclusions: • Cells were not significantly affected by exposure to empty or iron filled carbon nanotubes to concentrations up to 100 µg/ml. • These results indicate that the iron filling is well encapsulated inside of the nanotubes, leading to no major toxic effects. • Further studies will focus on the heating efficieny of cells and tissues incubated with iron filled CNTs. Evaluation after 96h Evaluation after 24h Evaluation after 48h Incubation with CNTs (4h) Cell Seeding Figure 4. Proliferation of PC-3 cells after exposure to empty and iron filled CNTs as evaluated by cell counting.